Article Summary 26/04/25
Article title
A cell-permeable nanobody to restore F508del cystic fibrosis transmembrane conductance regulator activity
Journal
Nature Chemical Biology
Tags
Nanobody, Cell permeable peptides
Introduction
Cystic fibrosis (CF) is a genetic disorder primarily caused by mutations in CFTR gene. The most prevalent mutation, F508del, leads to protein misfolding, and consequently, loss of chloride channel function. While small-molecule CFTR modulators, such as the triple-combination therapy elexacaftor/tezacaftor/ivacaftor (ETI), have improved clinical outcomes, they often provide only partial functional correction, highlighting an unmet therapeutic need.
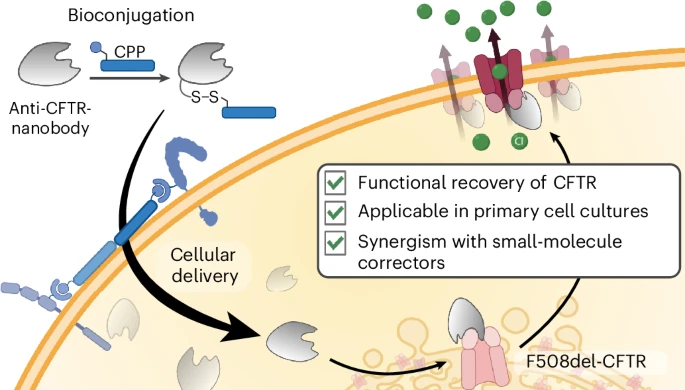
Nanobodies, represent a promising class of biologics due to their small size, high stability, and specificity. A previously identified nanobody, NB1, has been shown to act as a chaperone, stabilizing the F508del-CFTR protein in vitro and restoring its function. However, a major limitation for therapeutic application is the inefficient delivery of macromolecules like nanobodies into the cytosol to reach intracellular targets like CFTR. Cell-penetrating peptides (CPPs) offer a potential strategy to overcome this barrier by facilitating the cytosolic delivery of conjugated cargoes.
Thus, the central scientific question addressed in this study is whether a cell-permeable nanobody can be engineered to effectively rescue the function of misfolded F508del-CFTR within cells, potentially synergizing with existing therapies to achieve more complete functional restoration.
This work
This study designed, synthesized, and functionally validated a cell-permeable nanobody to restore F508del-CFTR activity. The core achievements are outlined below:
- Design and Synthesis of a Cell-Permeable Nanobody: The known CFTR-binding nanobody NB1 was conjugated to FITC and coupled to the arginine-rich cell-penetrating peptide R10 using a disulfide linkage strategy, creating a cell-permeable construct.
- Demonstration of Efficient Cytosolic Delivery and Target Engagement: the study demonstrated that NB1 was efficiently delivered into the cytoplasm of various cell lines and, critically, into highly differentiated primary human airway epithelial cultures derived from CF patients. The nanobody retained its ability to bind intracellular F508del-CFTR, as evidenced by co-localization studies.
- Rescue of F508del-CFTR Function in Model Cell Lines: Increased levels of the mature, complex-glycosylated Band C form of CFTR, indicating improved maturation and trafficking. Functional assays, including transepithelial short-circuit current measurements, confirmed the rescue of chloride channel activity
- Synergistic Functional Rescue in CF Patient-Derived Cells: The most significant finding was achieved in primary airway cultures from CF patients. The cell-permeable NB1-R10 nanobody not only entered cells but also synergized with the clinically approved small-molecule correctors in the ETI triple therapy. The combination therapy, targeting the NBD1 domain (via the nanobody) and other regions of CFTR (via small molecules), restored F508del-CFTR function to near-normal levels. This represents the first successful demonstration of CPP-mediated protein delivery and functional rescue by a nanobody in primary human cells from CF patients.
(Generated by DS v3.2.)
doi
10.1038/s41589-026-02199-w