Article Summary 26/03/07
Article title
High-throughput ligand diversification to discover chemical inducers of proximity
Journal
Nature Chemical Biology
Tags
Molecular glue; SuFEX; drug screening
Introduction
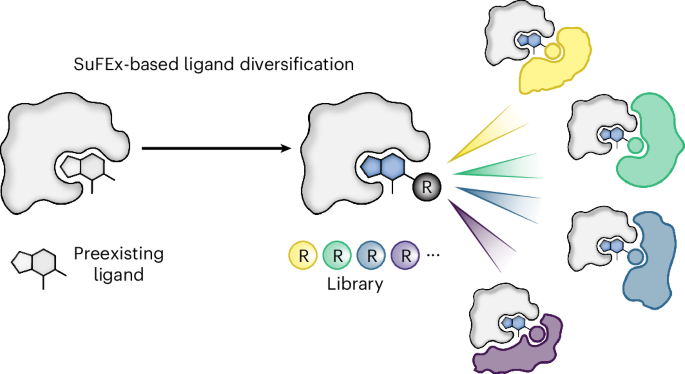
Chemical inducers of proximity (CIPs), such as proteolysis-targeting chimeras (PROTACs) and molecular glues, represent a powerful class of small molecules capable of rewiring cellular biochemistry by stabilizing proximity between target proteins. While rational design strategies have accelerated the discovery of heterobifunctional CIPs like PROTACs, the identification of molecular glues has historically relied on serendipity, lacking systematic discovery approaches. Molecular glues often share structural similarities with protein binders, raising the hypothesis that systematic diversification of existing ligands could yield novel glue-type CIPs.
This work
This study addresses the key scientific question: Can high-throughput ligand diversification serve as a systematic strategy to discover new molecular glue CIPs? The authors employed sulfur(VI) fluoride exchange (SuFEx)-based high-throughput chemistry (HTC) to install 3,163 structurally diverse building blocks onto known ligands for two targets: ENL (eleven-nineteen leukemia), a transcriptional regulator implicated in leukemia, and BRD4 (bromodomain-containing protein 4). Screening this library for degrader activity led to the discovery of dHTC1, an ENL degrader that functions as a molecular glue by recruiting the CRL4CRBN E3 ligase complex. Furthermore, for BRD4, they identified dHTC3, a molecular glue that selectively dimerizes BRD4 bromodomain 1 with the previously inaccessible E3 ligase FBXO3.
This work establishes HTC as a facile and potentially generalizable tool for the systematic discovery of novel CIPs and new effectors for proximity pharmacology, moving beyond reliance on chance findings towards a more prospective exploration of chemical space.
While this study demonstrates the potential of high-throughput ligand diversification as a systematic strategy for discovering novel chemical inducers of proximity (CIPs), it also highlights several limitations. First, the approach is fundamentally a screening method rather than a rational design strategy, which may limit its predictive power and efficiency. Second, the generalizability of this high-throughput chemical (HTC) strategy across different target classes remains to be validated, as its success here with ENL and BRD4 does not guarantee broad applicability. Finally, despite the identification of a promising degrader (dHTC1) for the important leukemia target ENL, the study lacks validation in animal disease models, which is crucial for assessing the therapeutic potential and translational value of the discovered compounds.
(Generated by DS v3.2.)
doi
10.1038/s41589-025-02137-2